Originally published on The Green lens.
Growing up watching sci-fi movies and reading comics, we have all had discussions with friends about who is the most powerful character with the ability to beat villains and even other heroes. Likewise, we have also imagined what it would be like to be a superhero or a villain (no judgement!) with our inexhaustive googling leading to questions about the existence of other life forms or even mythical legends like the Horta (from Star Trek movies) and Sandman (from Spider-man comics) in reality.
Although unique, these creatures are silicon-based rather than the usual major 4 – carbon, hydrogen, nitrogen and oxygen.
Why is carbon called the “backbone” of life?
Life is carbon-based, meaning that it is found everywhere on Earth, from our atmosphere to the food we eat to the ocean we see and even the land we walk on. Not only externally, but we are made up of carbon and its compounds internally too.
18.5% of the entire mass of the human body is made of the periodic table’s sixth element
It is widespread within us as the smallest atom in the body like our DNA, the bodily cells, layers of skin and even the nutrients our body needs among others – making it the “backbone”. For other living organisms, the carbon element goes as far as providing energy through ATP release. But how is the element so readily a part of life’s structure and functioning?
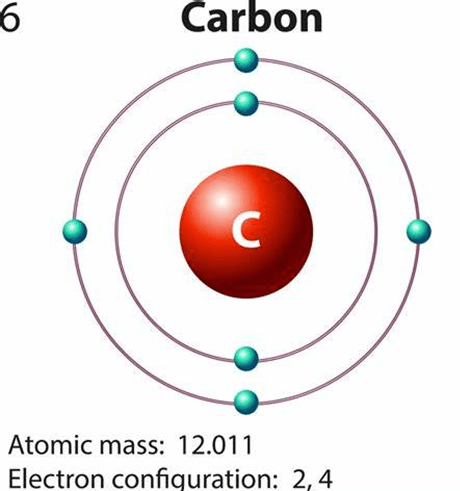
Carbon is versatile. Having a total of 6 electrons – 2 in its first orbit and 4 in its outermost orbit, it can share its electrons forming strong, stable and long-lasting covalent bonds with other elements. Diamond is one fine example of a carbon atom’s bond strength since it is the hardest element on the planet. Even in the presence of liquid mediums, the formations made by carbon with other elements remain unwavering. Owing to all of carbon’s flexible traits, it has easily been the most crucial component behind the lifeforms.
Could life be silicon-based and possibly replace carbon?
The concept of silicon biochemistry has been put forth by speculating scientists like Julius Scheiner, James Emerson Reynolds and JBS Haldane since the 1800s. The studies were based on silicon being an abundant element in nature. In fact, it is 135 times more abundant than carbon. The atom is even in the same column under the periodic table as carbon making it part of the carbon family yet scientists on Earth haven’t found silicon-based life. This is because like every other relationship, it all comes down to bonds, trust and stability!

Silicon has 4 unpaired electrons in its outer orbital, just like carbon. The main difference is silicon’s third i.e. outer orbit requires a total of 18 electrons whereas, for carbon, the second orbit is the outermost which requires only a total of 8 to be complete. When silicon bonds with other atoms including itself, the bonds formed are feeble due to a higher requirement of electrons. In contrast, carbon bonds are the strongest. Furthermore, silicon cannot make double or even triple bonds due to which the compounds formed lack strength.
Silicon molecules enjoy the perks of having low volatility and the ability to withstand intense temperatures (as high as 600K and possibly beyond). This is why silicon-based compounds are used in industrial lubricants and specialized products like U.S. missiles and satellites. However, in presence of liquid mediums like water, the atoms slowly start dissolving and reacting with hydrogen molecules causing the silicon bonds formed with other elements to break, unlike carbon.
Another important statement for silicon-based life to be unlikely is that the survival of life on Earth requires oxygen. When carbon reacts with oxygen it forms CO2 which is crucial for every living being. But when it comes to silicon, the solid SiO2 – basically sand – is formed which would look more like a Golem from the Jewish folklore (if shaped properly) and would be impossible to breathe out (obviously!).

Despite the statements put forth about carbon triumphing over silicon for life forms, perhaps silicon-based extra-terrestrial life may exist on other planets (even in other dimensions and within the multiverse) and even in odd areas of the Earth remaining undetected. Yet, if there are silicon-based life forms the evidence points towards rather lower forms of life instead of the intelligent complex creatures that inhabit the Earth.
References
- Does silicon-based life exist? | www.the-ies.org
- FAQ: Why is carbon the basis of life? – Answers to questions for all occasions (caagesf.org)
- CHAPTER 4 CARBON AND THE MOLECULAR DIVERSITY OF LIFE (reicheltscience.com)
- Why are carbon based molecules the foundation of life? (askinglot.com)
- Cosmic Evolution – Future (harvard.edu)
- Silicon-based life forms – Big Chemical Encyclopedia (chempedia.info)
- silicon-based life (daviddarling.info)
Discover more from Eco-intelligent
Subscribe to get the latest posts sent to your email.

[…] Image from eco-intelligent.com. […]
LikeLike